Description
Buy Epsom salt (Epsom salt, magnesium sulfate, magnesium salt)
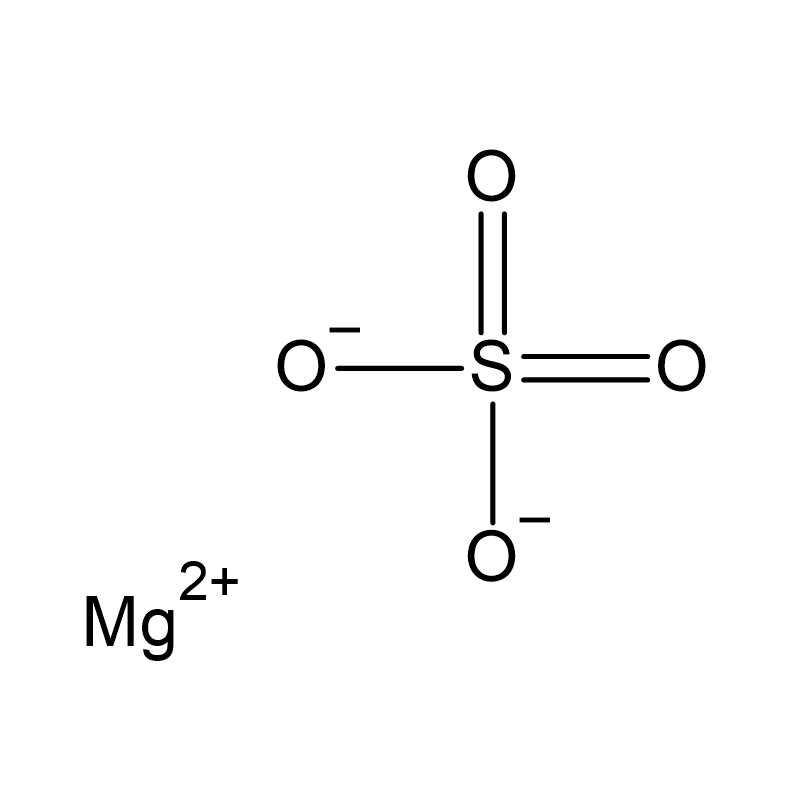
Looking to buy Epsom salt for professional use? This inorganic compound with the formula MgSO₄ is also known as Epsom salts or magnesium sulfate. It usually occurs as a white crystalline powder or colorless crystals. In its hydrated form, such as the heptahydrate, it has a wide range of applications. In industry and laboratories, it is used as a drying agent, additive, or reagent. In agriculture, it serves as a source of magnesium and sulfur to support crop growth.
Applications of Epsom salt in agriculture, laboratories and industry
In agriculture, Epsom salt is used to increase magnesium and sulfur levels in the soil. Due to its excellent water solubility, the compound decomposes into magnesium and sulfate ions upon dissolution. It is often used for magnesium-requiring crops such as potatoes, tomatoes, carrots, peppers, and citrus fruits like limes. An additional advantage of Epsom salt is its pH neutrality, which prevents damage to soil structure and eliminates the need for stabilizers.
In chemistry, the substance is often used as a desiccant in organic synthesis because of its strong water-absorbing properties. The hydrated residue can be easily removed after use by filtration, decantation, or, if the boiling point of the solution is low enough, by distillation. Other suitable desiccants are sodium sulfate and calcium sulfate .
The substance can also be used in the production of specific sustainable cement.
It is also used to maintain magnesium concentrations in certain water solutions, for example, in laboratory analyses. Regular monitoring of magnesium levels is recommended.
Storage and use
Store magnesium sulfate in a tightly closed container in a dry, well-ventilated place. Avoid contact with moisture, as it is highly hygroscopic. Avoid storage near strong acids and bases, or substances that react exothermically with water. Use only in suitable professional applications such as laboratories or industrial facilities.
Frequently Asked Questions (FAQ)
Is Epsom salt the same as Epsom salts?
Yes, both names refer to magnesium sulfate heptahydrate.
Is magnesium sulfate the same as Epsom salts?
Yes, magnesium sulfate is also known as Epsom salts and also Epsom salts. It is a versatile chemical compound with the formula MgSO₄ and is used in various applications such as chemical processing.
Can I use Epsom salt in horticulture?
Yes, it is often used as a source of magnesium in cases of magnesium deficiency in soil or plants.
What is the difference between anhydrate and heptahydrate?
The anhydrate contains no water of crystallization and is drier; the heptahydrate contains seven molecules of water per formula unit.
Is this substance soluble in water?
Yes, the heptahydrate dissolves well in water.
Learn more
For additional information on the chemical properties and uses of magnesium sulfate, please refer to the following source: PubChem – Magnesium Sulfate










Reviews
There are no reviews yet