Description
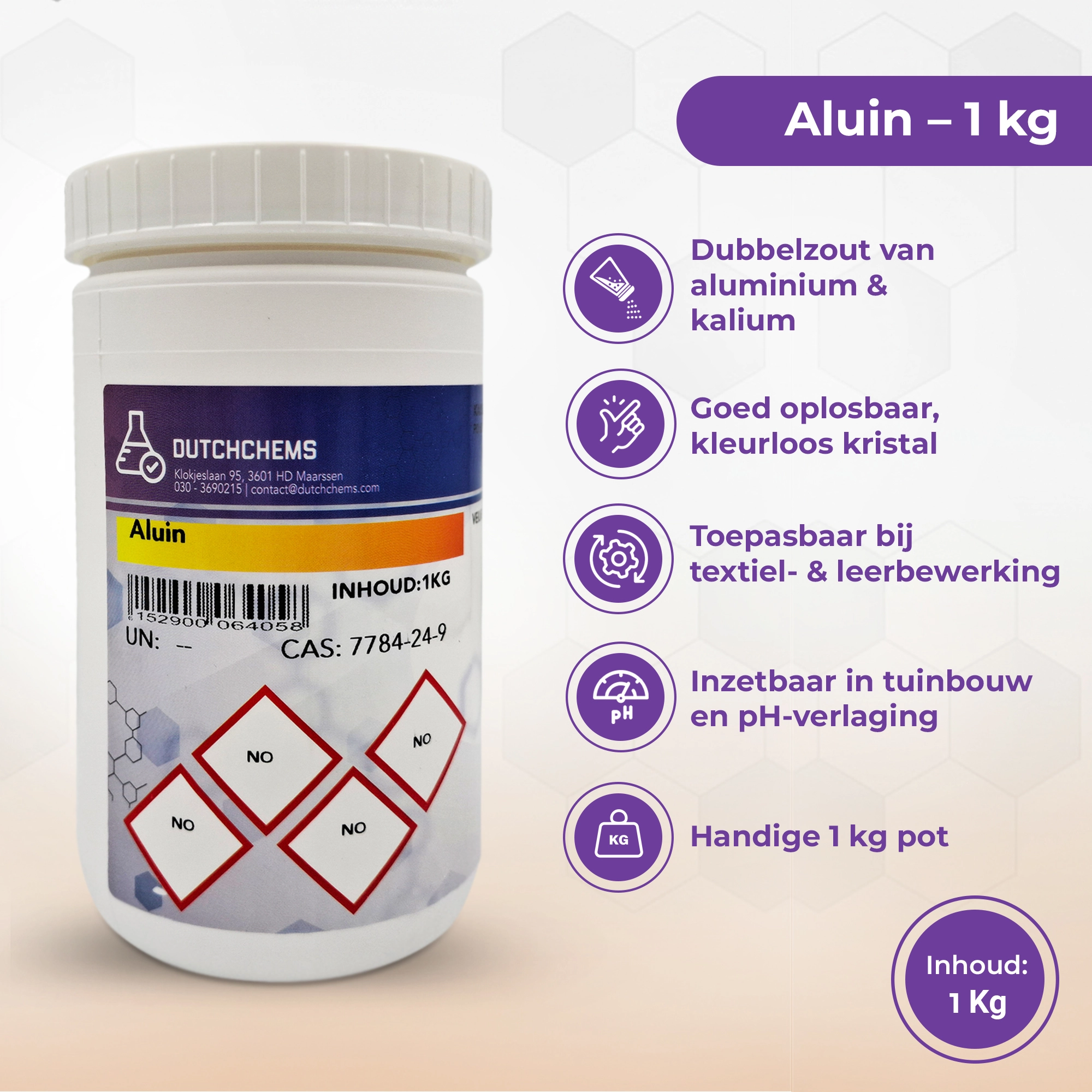
Buy Alum (Potassium Aluminum Sulfate, Aluminum Potassium Sulfate)
Want to buy alum? Alum is a naturally occurring chemical compound, a so-called double salt consisting of aluminum, sulfate, and an alkali ion such as potassium. The most commonly used form is potassium alum with the chemical formula KAl(SO₄)₂·12H₂O. It appears as a colorless to white crystalline powder that dissolves easily in water. Alum is often used in textile processing. With a purity of 97% and higher, it is suitable for professional and technical use.
Product Specifications
- Product name: Alum
- Also known as: Potassium Aluminum Sulfate, Aluminum Potassium Sulfate
- CAS number: 7784-24-9
- Chemical formula: KAl(SO₄)₂·12H₂O
- Purity / quality: 97% – 99%
- Form: White to colorless crystalline powder
- Application: Flocculant, textile industry, laboratories
What is Alum used for?
Thanks to its unique chemical properties, alum plays a vital role in a variety of traditional and modern processes. Traditionally, it was used in papermaking to improve the paper’s properties. It was added to paper pulp to strengthen its structure and increase ink resistance. This reduced the fibers’ ink absorption, resulting in a sharper print image. Although this application is less common today, it remains a historically important ingredient in the development of the paper industry.
Potassium alum is an essential tanning agent. It is used to preserve and soften hides. Its astringent properties stabilize the fibers in the hide, making the leather more supple and resistant to wear. It is primarily used in the production of white leather, where it produces a smooth, light finish without dark discoloration.
Alum is widely used as a mordant in textile dyeing. It improves the adhesion of dyes to the fibers, thus ensuring better colorfastness. Pre-treating fabrics with this substance optimizes the chemical reaction between the fibers and the dye, resulting in more intense and durable colors.
Alum is also widely used in water purification. It acts as a coagulant and helps remove dirt particles and other impurities from water. Small particles clump together into larger flakes, which then settle to the bottom or can be easily filtered out. This process makes alum a commonly used substance in the treatment of process and industrial water.
In horticulture, alum is valued for its ability to lower soil pH. Acid-loving plants, such as hydrangeas, benefit from the addition of this substance to the soil. By making the soil more acidic, a favorable growing environment is created for these plants, which is reflected in the more intense colors of hydrangeas.
Chemical properties
- Density: 1.76 g/cm³ (solid powder)
- Melting point: 92 °C (water molecules escape; decomposition around 200 °C)
- Solubility:
- In water: 141 g/L at 20 °C (readily soluble)
- In ethanol: Insoluble
- pH solution: Weakly acidic (pH ±3.5 in 10% aqueous solution)
- Molecular weight: 474.39 g/mol
Storage and use
Store alum in a dry, tightly closed container, preferably at room temperature in a well-ventilated area. Protect from moisture to prevent clumping or unwanted dissolution. Keep away from strongly alkaline substances. Use only in suitable professional environments and adjust dosage and handling to the specific application and process conditions.
Frequently Asked Questions (FAQ)
Is alum suitable for laboratory use?
Yes, it is used in laboratories for controlled chemical and analytical processes.
How should alum be stored?
Dry, well closed and protected from moisture.
What form does it come in?
Usually as crystalline powder or solid crystals depending on packaging and application.
Learn more
For additional information on the chemical properties and applications of Alum, please refer to the following source: PubChem – Alum (Potassium Alum)












1 review for Alum – Potassium aluminum sulfate – Potassium alum
There are no reviews yet