What is Formic Acid?
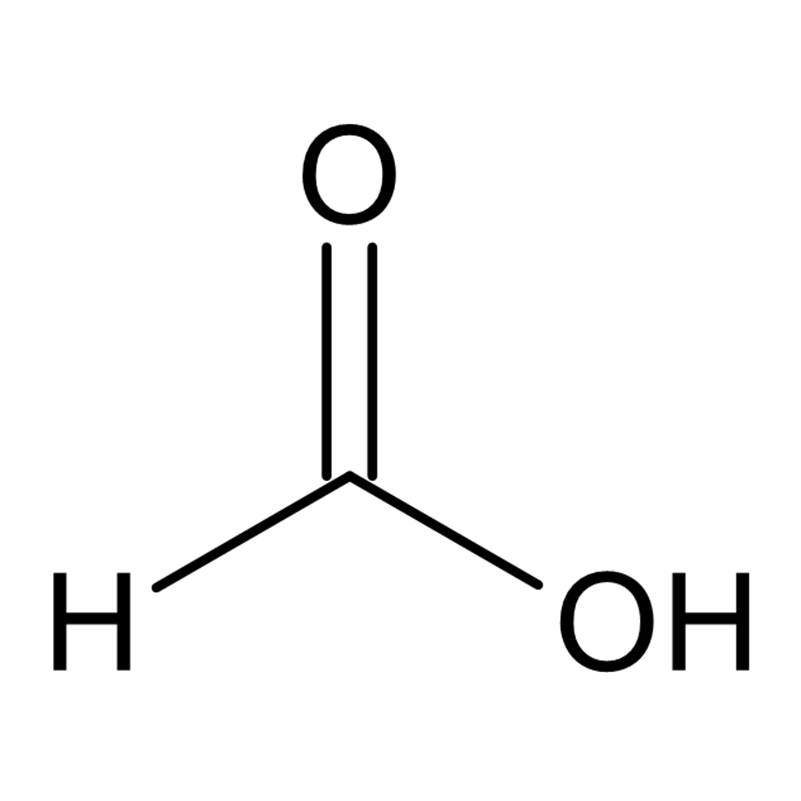
Formic acid, with the chemical formula HCOOH, is the simplest carboxylic acid. It is a colorless liquid with a sharp, pungent odor. The name is derived from the fact that the acid was originally isolated from ants.
What do you use formic acid for?
Formic acid has a wide range of industrial and laboratory applications, including:
- Leather production: Used in tanning leather.
- Textile industry: Used for dyeing and finishing textiles.
- Agriculture: As a remedy for pests and parasites.
- Preservative: In food storage and processing.
- Chemical research: As a reagent in various laboratory applications.
Dangers of Formic Acid
Formic acid can cause irritation to the skin, eyes and respiratory tract upon contact. If inhaled or swallowed, it can have toxic effects. Therefore, it is important to handle with care and use personal protective equipment.
Other common areas of application:
- Production of rubber and plastics.
- As a coagulant in the production of latex.
FREQUENTLY ASKED QUESTIONS
- Is formic acid dangerous?
- Yes, formic acid can cause irritation on direct contact and is harmful if inhaled or swallowed. Always ensure adequate protection.
- Why is it called formic acid?
- The acid was originally isolated from ants, hence the name.
- Where can I find more information about Formic Acid?
- More details about formic acid can be found on https://pubchem.ncbi.nlm.nih.gov/compound/Formic-Acid
Where can I buy high quality formic acid?
Are you looking for top quality formic acid? DutchChems has what you need. Order easily and enjoy our fast delivery. Please note the special shipping requirements depending on the application.



















Reviews
There are no reviews yet