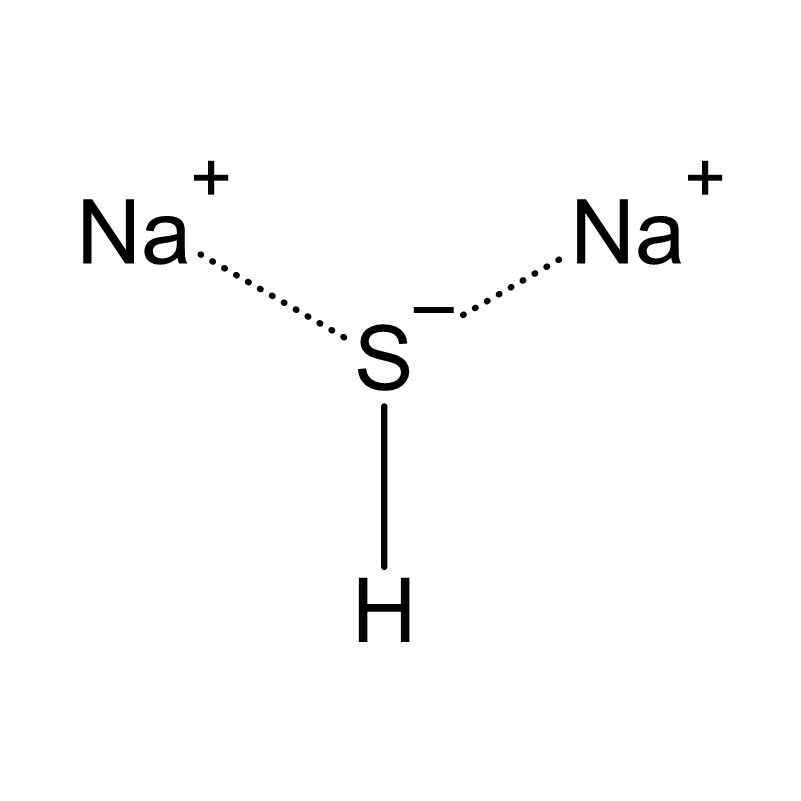
What is Sodium Sulfide?
Sodium sulfide (Na2S) is a yellow to reddish-brown crystalline salt that excels in its reductive properties. Due to its characteristic sulfurous smell and various industrial applications, it is a valuable chemical in various sectors.
What do you use Sodium Sulfide for?
Sodium sulfide has several applications:
- Leather industry: When depilating animal skins prior to tanning.
- Textile industry: For removing excess dyes from dyed materials.
- Mining: As a flotation agent in the processing of mineral ores.
Properties of Sodium Sulfide
Sodium sulfide is highly soluble in water and can release toxic gas (hydrogen sulfide) when exposed to acidic conditions. Caution should be exercised when storing and using.
Other common areas of application:
- In the pulp and paper industry for cooking wood pulp.
- As a gear unit in the photography industry.
FREQUENTLY ASKED QUESTIONS
- How should I safely store Sodium Sulfide? It should be stored in a dry, cool and well-ventilated place, in a tightly sealed container, away from acids and oxidizing agents.
- Is Sodium Sulfide Dangerous? Yes, contact with the skin and eyes can cause burns, and exposure to acidic conditions can produce toxic gas. Always follow safety guidelines when using it.
- Where can I find more information about Sodium Sulfide? More information can be found in the safety data sheets offered, or at: Pubchem.
Where can I buy Sodium Sulfide?
Looking for Sodium Sulfide for the leather, textile or mining industry? DutchChems offers top quality sodium sulfide for various professional needs.




















Reviews
There are no reviews yet